
Invention Spotlight: Patent Issued for Personalized Motion-Gated Coronary CTA Scanning Systems and Methods
Newly issued patent enables patient-specific coronary CTA imaging by predicting cardiac motion to improve image quality and accuracy.
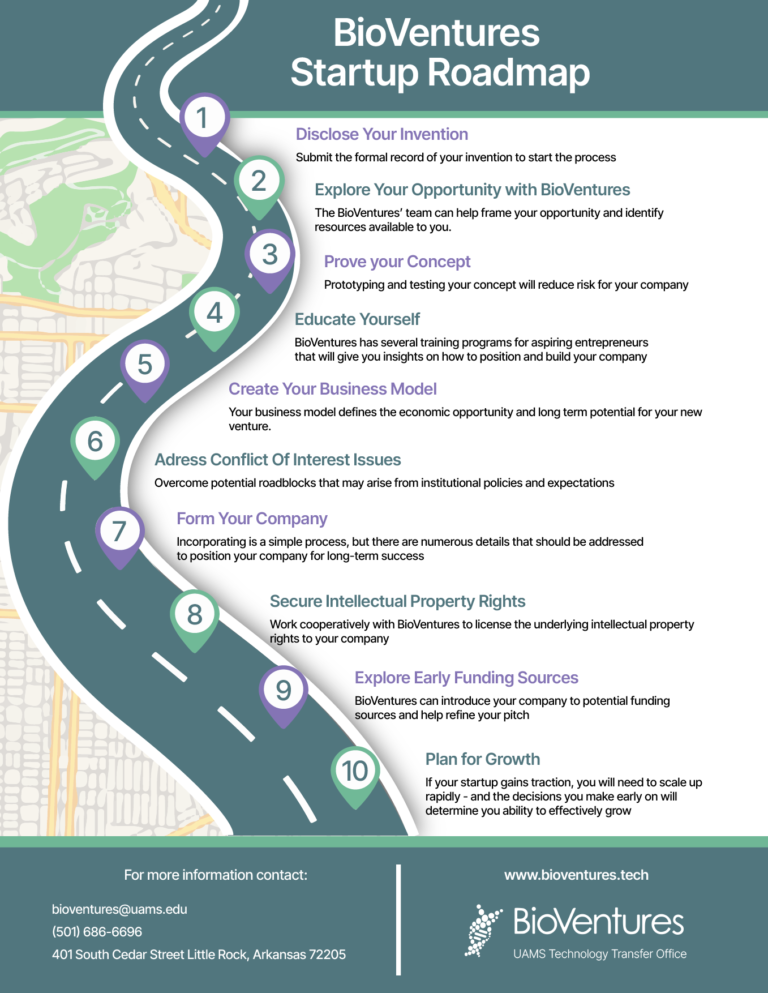
The invention disclosure process can be overwhelming at times, which is why we have broken down the step by step process for you. Here you can read the timeline in a simple and clear way. If you are an inventor, click here to read a more detailed description of the process.
Submit the invention disclosure form stating the current status of your invention.
Our team will complete the TAR, a structured summative document regarding your invention. This includes a snapshot of colesely related inventions, and preliminary market analysis.
The slide deck helps us focus the technology assessment report even more and allows us to send the information to individuals in the industry to help us assess the technology.
The Bioventures team preset the technology to the Committee with the collaboration of the inventor.
After the Patent and Copyright Committee meet, there are 3 possible determinations.
To start the process, the first step is to fill out the Invention Disclosure Questionnaire. Log in to Wellspring Sophia and click “Login through UAMS”.

Newly issued patent enables patient-specific coronary CTA imaging by predicting cardiac motion to improve image quality and accuracy.

UA Little Rock will host Pitches & Demos for the AI Hackathon & HealthTech Startup Week on Friday, June 12, from 2:00 to 5:00 p.m. at the Engineering & Information Technology Auditorium.

Dr. Anthony Gunderman discussed how engineering and robotics can help address clinical needs during a recent UAMS Research & Innovation seminar.
Stay tuned with BioVentures’ newsletter for inventors: news, events, resources and more!